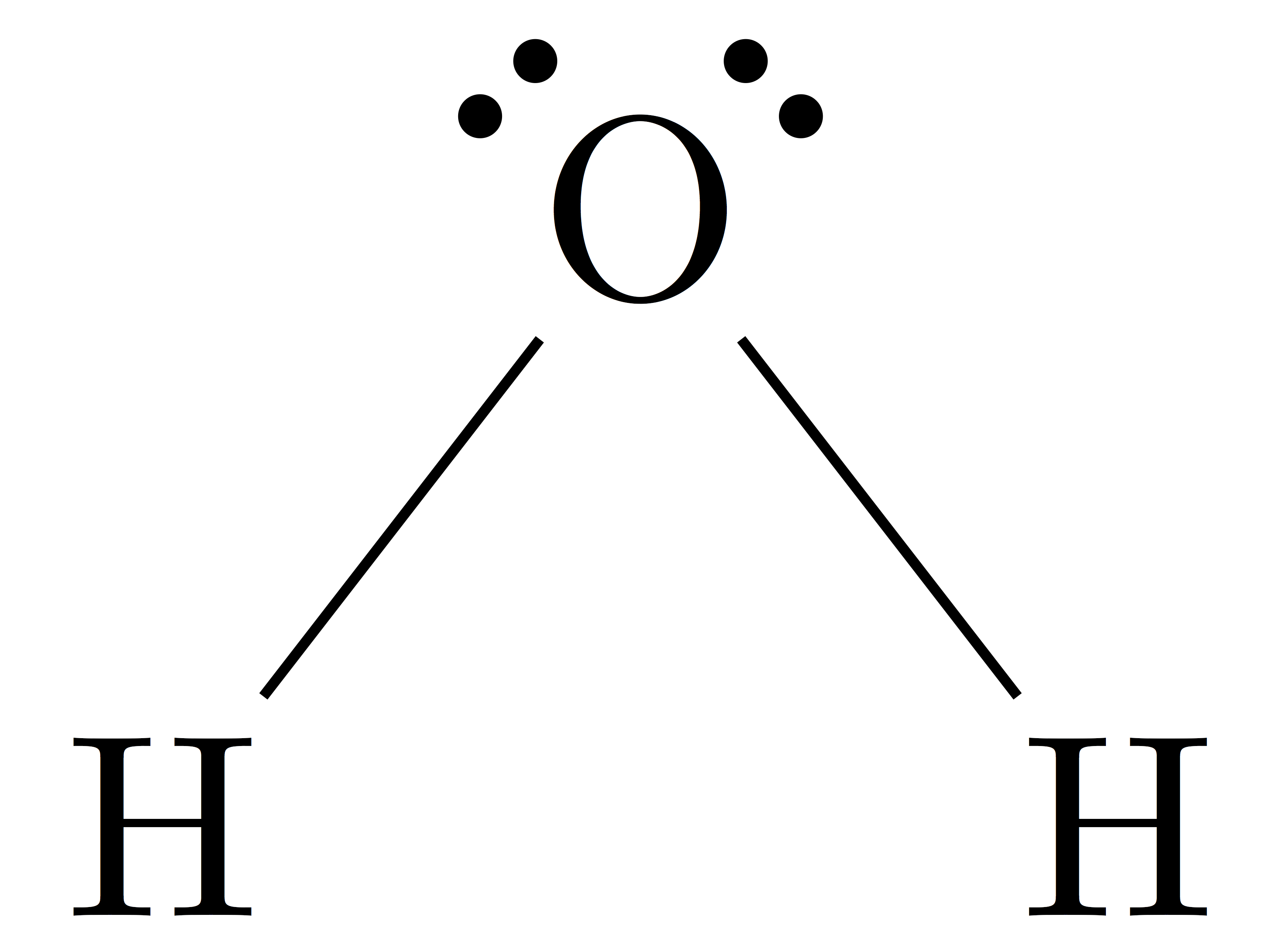
This molecule has three regions of high electron density: two single bonds and one double bond. Lone pair > triple bond > double bond > single bondĬonsider formaldehyde, H 2CO, which is used as a preservative for biological and anatomical specimens. VSEPR theory predicts distortions from ideal angles by approximating an order of electron-region repulsions, where: (c) The actual bond angles deviate slightly from the idealized angles because a lone pair causes the remaining bond angles to compress, causing the HNH angle to be slightly smaller than 109.5°. (b) The molecular geometry is a triangular-based pyramid, with H atoms at the base of the pyramid. (a) The electron-region geometry for NH 3 is tetrahedral with one lone pair and three single bonds. One of these regions, however, is a lone pair, which is not included in the molecular geometry, and this lone pair influences the shape of the molecule (Figure 2). The two geometries are the same only when there are no lone pairs around the central atom.įor example, ammonia, NH 3, has four electron pairs associated with the central nitrogen atom, and thus has a tetrahedral electron-region geometry.  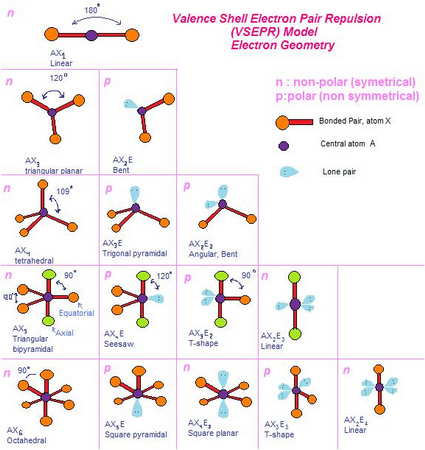
The molecular geometry (or molecular structure) describes the location of the atoms (that is, atomic nuclei and core electrons), not the valence electrons. The electron-region geometry describes all regions where valence electrons are located, bonds as well as lone pairs. D11.4 Electron-Region Geometry versus Molecular Geometry A molecular model and a Lewis structure for methane look like this:Īny molecule with five electron regions around the central atoms with no lone pairs will be trigonal bipyramidal. The H atoms are at the four corners of a tetrahedron. Each C–H bond is 109.5° from all three other C–H bonds. The largest possible angle between electron pairs is 109.5°, which is called a tetrahedral angle. The basic electron-pair geometries predicted by VSEPR theory maximize angles between regions of electron density (bonds or lone pairs).įor example, methane, CH 4, which is the major component of natural gas, has four bonding pairs of electrons around the central carbon atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
